Every medication prescribed in a private consultation carries a Summary of Product Characteristics (SmPC) — the definitive reference document for that medicine’s safe use. For pharmacists and prescribers offering private prescribing services, SmPC-guided consultations are not just best practice — they are a fundamental requirement for patient safety and regulatory compliance.
This guide explains what SmPCs are, why they matter for private prescribing, and how software can integrate SmPC guidance directly into the consultation workflow.
What Is an SmPC?
The MHRA (Medicines and Healthcare products Regulatory Agency) approves the Summary of Product Characteristics for every licensed medicine in the UK. It contains:
- Therapeutic indications — what the medicine is licensed to treat
- Posology and method of administration — dosage, frequency, and route
- Contraindications — conditions or circumstances where the medicine must not be used
- Special warnings and precautions — risks to be aware of during treatment
- Interactions — other medicines, foods, or substances that may interact
- Pregnancy and lactation — safety information for pregnant or breastfeeding patients
- Undesirable effects — known side effects and their frequency
- Overdose — symptoms and management of overdose
The SmPC is the gold standard reference for prescribing decisions. It differs from the Patient Information Leaflet (PIL), which is a simplified version written for patients.
Why SmPCs Matter for Private Prescribing
Clinical Governance
When you prescribe privately, you take full clinical responsibility for that prescribing decision. The SmPC provides the evidence base for your decision. If a prescribing decision is ever questioned — by the patient, their GP, or the GPhC — the SmPC is the reference against which your decision will be measured.
Patient Safety
SmPCs contain critical safety information that may not be immediately recalled from memory. Even experienced prescribers benefit from systematic SmPC checks, particularly for:
- Medicines they prescribe infrequently
- Patients with multiple comorbidities
- Patients on multiple medications (polypharmacy)
- Recently updated SmPCs with new safety warnings
- Off-label prescribing decisions that require documented justification
Regulatory Compliance
GPhC standards require prescribers to make evidence-based prescribing decisions and demonstrate that they have considered relevant safety information. SmPC-guided consultations provide documented evidence of this consideration.
The Problem with Manual SmPC Checks
In a busy pharmacy or clinic, the reality of manual SmPC checking is challenging:
- Time pressure — looking up SmPCs on the MHRA website during a consultation takes 3–5 minutes per medication
- Inconsistency — under time pressure, checks may be skipped for familiar medications
- No audit trail — there is no record that the SmPC was reviewed
- Version control — SmPCs are updated regularly; are you checking the current version?
- Multi-medication complexity — checking interactions across multiple medications manually is error-prone
How Software Integrates SmPC Guidance

Modern e-prescribing software can integrate SmPC information directly into the consultation and prescribing workflow:
Automated Contraindication Alerts
When a prescriber selects a medication, the system automatically checks the patient’s recorded conditions against the SmPC contraindications. If it finds a contraindication, it alerts the prescriber immediately before generating the prescription.
Drug Interaction Checking
The system cross-references the proposed medication against the patient’s current medication list, flagging interactions classified in the SmPC. Interactions are typically categorised by severity: critical (do not co-prescribe), significant (monitor closely), or minor (be aware).
Dosage Guidance
The system presents SmPC-derived dosage ranges during prescribing, helping prescribers select appropriate doses.. The system can flag doses outside the licensed range, prompting the prescriber to document their clinical rationale if proceeding.
Pregnancy and Lactation Warnings
For patients flagged as pregnant or breastfeeding, the system automatically displays the SmPC pregnancy and lactation guidance for any considered medication.
Patient Counselling Points
The system can automatically include key SmPC information in patient-facing documentation, such as common side effects to watch for, important interactions with food or alcohol, and when to seek urgent medical advice.
Building SmPC Checks into Your Consultation Workflow
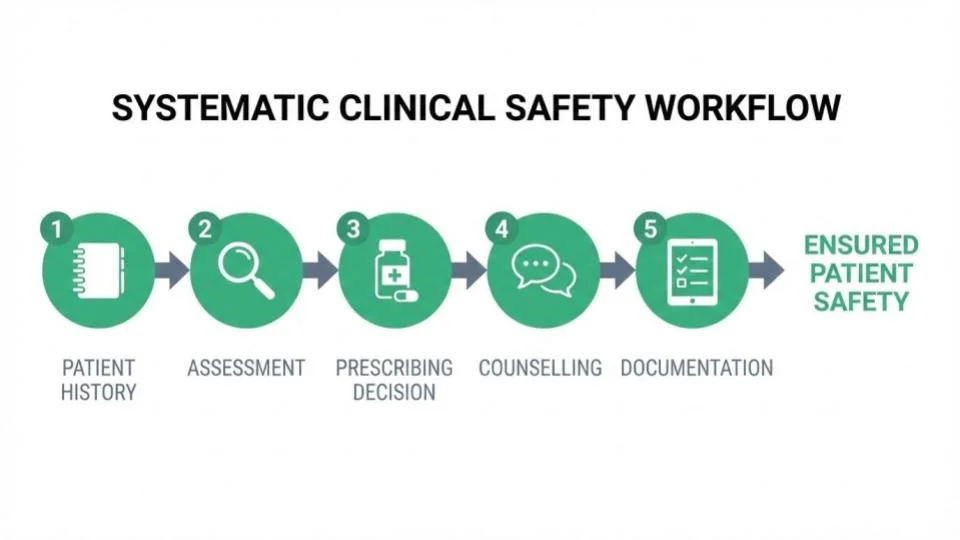
Step 1: Patient History
Capture complete medical history, current medications, allergies, and pregnancy/lactation status. This data feeds the automated safety checks throughout the consultation.
Step 2: Clinical Assessment
Use structured consultation templates that prompt for condition-specific information. The template should align with the therapeutic indications in the relevant SmPC.
Step 3: Prescribing Decision
When selecting a medication, review the automated SmPC alerts. Document your clinical rationale, especially if overriding any alerts or prescribing outside licensed indications.
Step 4: Patient Counselling
Share relevant SmPC information with the patient: expected effects, common side effects, important precautions, and when to seek help. Document that counselling was provided.
Step 5: Documentation
The consultation record should demonstrate that SmPC-relevant safety checks were performed. With integrated software, this documentation is automatic — creating an audit trail that satisfies GPhC inspection requirements.
SmPC Considerations for Common Private Services
Weight Management (GLP-1 Agonists)
SmPCs for semaglutide and liraglutide contain important guidance on dose titration schedules, pancreatitis warnings, thyroid cancer precautions, and pregnancy contraindications. Weight management clinics must build these checks into every consultation.
Sexual Health
The system must systematically check medications like sildenafil for critical cardiovascular contraindications and nitrate interactions. SmPC-guided prescribing prevents potentially dangerous interactions.
Dermatology
Isotretinoin has extensive SmPC requirements including pregnancy prevention programmes, liver function monitoring, and mental health warnings. Software should enforce these checkpoints.
Travel Health
Antimalarial medications have specific SmPC guidance on timing, food interactions, and contraindications. Automated checks ensure the right antimalarial is selected for each patient’s profile.
The Business Case for SmPC Integration
Beyond patient safety, SmPC-guided consultations benefit your business:
- Reduced clinical risk — systematic safety checks reduce the likelihood of adverse events and associated liability
- Faster consultations — automated checks are faster than manual SmPC lookups
- Staff confidence — newer prescribers feel more confident with built-in safety nets
- Inspection readiness — automatic audit trails satisfy regulatory requirements
- Patient trust — demonstrating thorough safety checks builds patient confidence in your service
Key Takeaways
- SmPCs are the definitive reference for safe prescribing — every private prescribing decision should be SmPC-informed
- Manual SmPC checking is time-consuming, inconsistent, and creates no audit trail
- Integrated software automates contraindication checks, interaction alerts, and dosage guidance
- SmPC-guided consultations protect patients, prescribers, and your business
- Automatic documentation satisfies GPhC requirements for evidence-based prescribing
- RxSure integrates safety checks and clinical decision support into the electronic prescribing workflow
Ready to build SmPC safety into every consultation? Start your free 3-month RxSure trial — safer prescribing from day one.