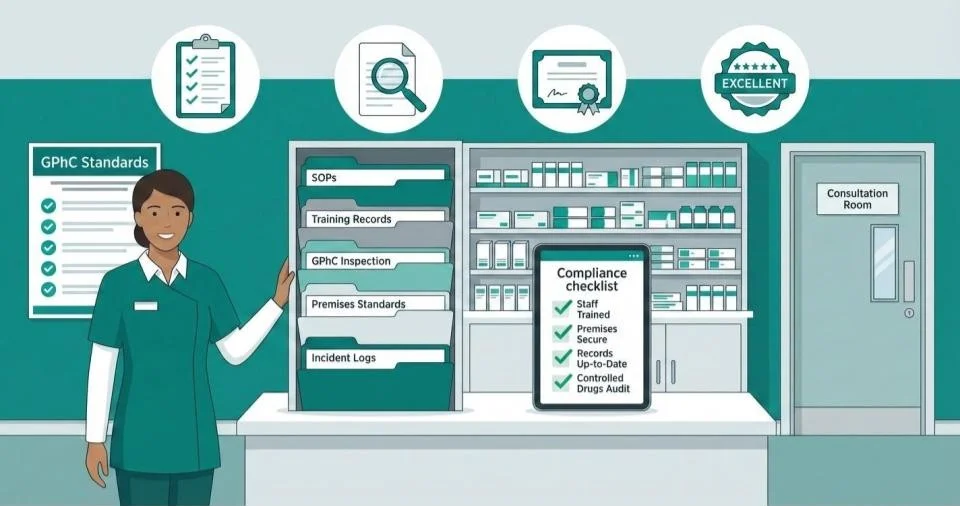
The General Pharmaceutical Council approach to pharmacy inspection has evolved significantly. The introduction of the focused inspection model in January 2025 fundamentally changed how pharmacies are assessed, what evidence is required, and how outcomes are determined.
Understanding these changes is not just about passing inspections. It is about embedding the standards that protect patients and support professional practice into your pharmacy’s daily operations.
This comprehensive guide outlines the current inspection framework in detail, clearly explaining the evidence requirements for each core standard. In addition, it offers practical preparation strategies designed to support ongoing compliance, ensuring the pharmacy remains confident and well-prepared—whether an inspection is imminent or still months away.

The Focused Inspection Model Explained
Shifting from Comprehensive to Focused
Previously, GPhC inspections attempted to assess all aspects of pharmacy operations in a single visit. This comprehensive approach, while thorough, often felt overwhelming and did not always allow deep examination of critical areas.

The focused inspection model concentrates inspector attention on 18 core standards identified as most critical to patient safety and professional practice. This does not mean other standards are unimportant.
The focused inspection model concentrates inspector attention on 18 core standards identified as most critical to patient safety and professional practice. This does not mean other standards are unimportant. Pharmacies must still meet all requirements but inspections prioritise the areas where failures pose greatest risk.

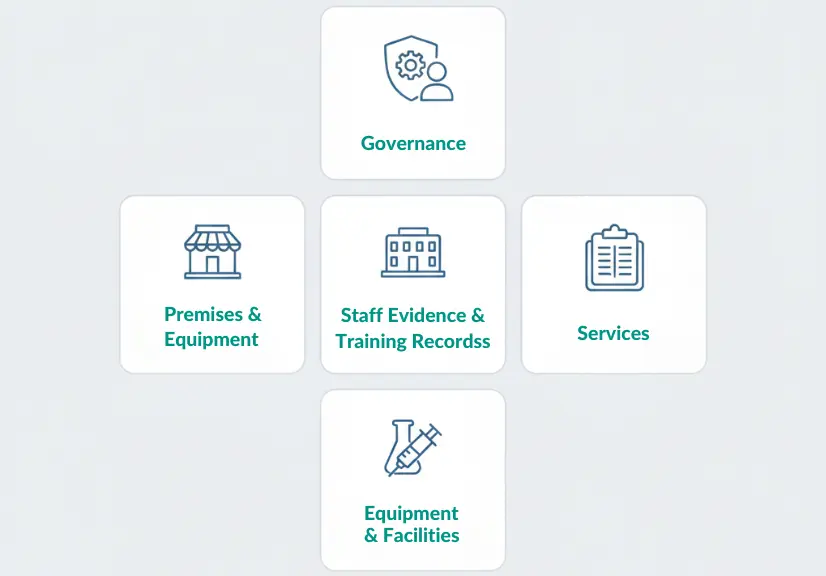
The 18 Core Standards
The core standards fall under five principles:
Governance
- Identifying and managing risk
- Reviewing and monitoring the safety of services
- Appropriate indemnity arrangements
- Appropriate insurance arrangements
Staff
- Sufficient staff for safe and effective services
- Staff qualifications and competence
- Staff training and development
Premises and Equipment
- Premises suitable for services provided
- Equipment fit for purpose
- Medicines stored safely and securely
Services
- Safe and effective service delivery
- Medicines supplied safely
- Advice given accurately and appropriately
- Service user involvement in decisions
Equipment and Facilities
- Accurate records maintained
- Confidentiality protected
- Complaints handled appropriately
- Concerns and incidents reported
Inspection Outcomes
The focused model produces clearer outcomes:
- Standards met: All core standards satisfactorily evidenced
- Standards partly met: Some evidence of compliance with improvement needed
- Standards not met: Significant gaps requiring urgent action
- Improvement action plan: Required for partly met or not met standards
Evidence Requirements by Standard
Governance Evidence
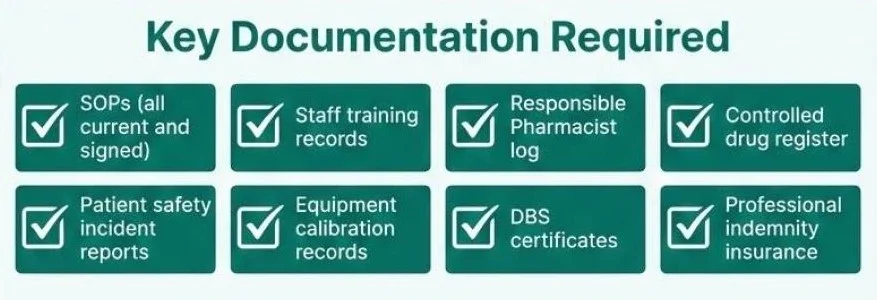
Inspectors examining governance will request:
- Risk assessments: Written assessments of key operational risks with mitigation strategies
- Audit records: Evidence of regular self-assessment and improvement actions
- Insurance certificates: Current professional indemnity and public liability documentation
- Incident logs: Records of errors, near misses, and actions taken
Staff Evidence
For staffing standards, prepare:
- Rota records: Evidence of appropriate skill mix at all times
- GPhC registration checks: Verification of current registration for all registrants
- Training records: Documented competency assessments and ongoing development
- DBS certificates: Current checks for all relevant staff
Premises Evidence
Premises and equipment evidence includes:
- Temperature logs: Continuous monitoring of medicine storage areas
- Calibration records: Evidence that measuring equipment is accurate
- Maintenance logs: Regular checks and repairs documented
- Cleaning schedules: Evidence of hygiene standards
Services Evidence
Service delivery evidence encompasses:
- SOPs: Written procedures for all services offered
- Patient records: Appropriate documentation of consultations
- Dispensing audit data: Error rates and improvement trends
- Patient feedback: Collection and response to comments
Practical Preparation Strategies
Continuous Readiness vs Last-Minute Panic
The most effective inspection preparation happens every day, not in a panic when notice arrives. Pharmacies with continuous compliance cultures find inspections straightforward because evidence is always current and accessible.

Key principles for continuous readiness:
- Maintain all documentation in real-time
- Address issues when they arise, not when inspectors ask
- Regular self-audit against the 18 core standards
- Team awareness of evidence locations and responsibilities
The 30-Day Preparation Plan
If you receive inspection notice then use this structured approach:
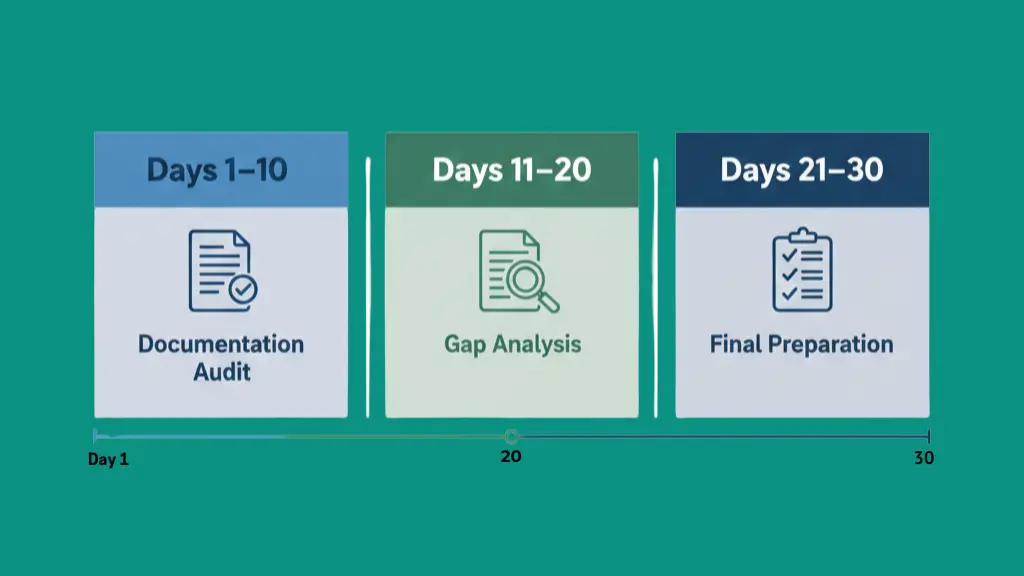
Documentation Audit: Days 1-10:
- Verify all certificates are current and accessible
- Update any expired or missing training records
- Ensure SOPs reflect current practice
- Check temperature and calibration logs are complete
Gap Analysis: Days 11-20
- Self-assess against each core standard
- Identify areas needing improvement
- Create action plan for any gaps
- Brief team on findings and actions
Final Preparation: Days 21-30
- Complete any outstanding actions
- Organise evidence files for easy access
- Team briefing on inspection process
- Practice articulating your pharmacy strengths
During the Inspection
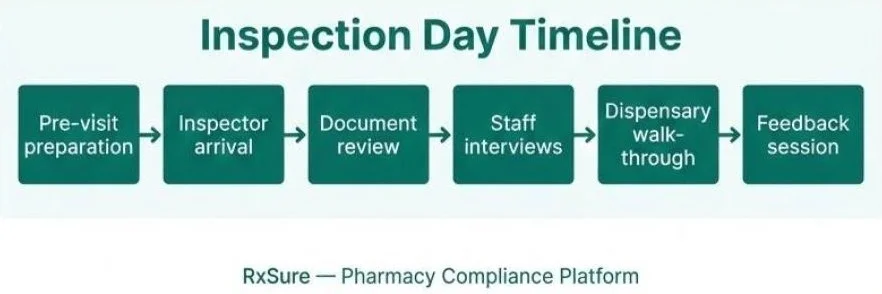
What to Expect
Typical inspection activities include:
- Initial discussion with responsible pharmacist
- Observation of dispensing and service delivery
- Staff interviews across different roles
- Documentation review
- Premises walkthrough
- Feedback discussion
Communication Tips
During inspector interactions:
- Be honest about challenges and how you address them
- Provide evidence proactively rather than defensively
- Involve team members appropriately
- Ask clarifying questions if unsure what is being requested
- Take notes for your own records
After the Inspection
Response to Findings
If improvement actions are required:
- Acknowledge findings constructively
- Create detailed action plan with timescales
- Assign clear ownership for each action
- Document completion evidence
- Submit response within required timeframe
Learning and Improvement
Every inspection is a learning opportunity:
- Team debrief on findings and feedback
- Identify systemic issues requiring broader change
- Update procedures based on inspector guidance
- Share learning across sites if multi-branch
Summary
Effective General Pharmaceutical Council (GPhC) inspection preparation should be embedded into daily pharmacy operations rather than approached as a one-time or periodic event. By consistently aligning practices with the 18 core standards, maintaining up-to-date and well-organised evidence, and cultivating a strong team culture of compliance, pharmacies position themselves not only to successfully meet inspection requirements but also to deliver consistently high standards of patient care.
Need help preparing for your GPhC inspection?
RxSure platform includes inspection preparation tools including compliance dashboards, evidence generation, and audit checklists aligned to GPhC core standards. Start your free trial and approach your next inspection with confidence.